- News & Event
-
General Information
In the last ten years, there has been a dramatic change in the global economic and scientific landscape. The emerging economies of India, China, Korea, Hong Kong, Taiwan, Thailand, Singapore, Malaysia and Japan have arisen into a strong economic block. Lately, the Middle Eastern Countries have also been undergoing a transformation in their perception and approach towards science and technology. Saudi Arabia, Qatar, Kuwait and Dubai are making huge investments to strengthen science and technology. Further, these countries are spending a significant proportion of their resources into building biotechnological capabilities. There is growing consensus that developments in biotechnology will have tremendous implications for every aspect of life on this planet including the food we eat, the clothes we wear, management and protection of our environment, reproduction and maintenance of biodiversity, and the prevention and management of such dreadful diseases as tuberculosis, HIV and cancer.
Asia-Pacific IMBN is a young organization with a mission to maintain and promote the development of scientific and technical excellence amongst scientists and institutions dedicated to molecular biology and genetic engineering research. A-IMBN recognizes that there are many international organizations working for goals similar to those of A-IMBN. Most notable among them are EMBO, HUGO, ICGEB, ISESCO, COMSTECH and TWAS. Since their interests coincide to form a complementarity, A-IMBN has attempted to develop sister linkages for mutually beneficial research and training programs; enter into partnership rather than competition; avoid duplication and wasteful expenditures. A-IMBN makes a judicious selection of choices and undertakes programs that would result into building individual/institutional capabilities, promote applications of molecular biology/biotechnology.
A-IMBN is particularly keen to develop linkages with Islamic countries/organizations and romote activities in the Islamic world for two reasons. Firstly, the countries in the Islamic world present a significant proportion of the world population. Secondly, the Islamic countries need to use biotechnological procedures to exploit their natural resources to accelerate the pace of economic growth. This is a corner stone of A-IMBN policies and this in view, A-IMBN and ISESCO are jointly developing a training course for young and initiated researchers in the Asia-Africa region to introduce them to Good Manufacturing Practice (GMP).
GMP is part of a quality system covering the manufacture and testing of active pharmaceutical ingredients, diagnostics, foods, pharmaceutical products and medical devices. GMPs are guidelines that outline the aspects of production and testing that can impact the quality of a product. Many countries have legislated that pharmaceutical and medical devices companies must follow GMP procedures, and have created their own GMP guidelines that correspond with their legislation. Although there are a number of them, all guidelines follow a few basic principles, which will be taught to the participants of the course.
The course will equip participants with GMP requirements, so they can design experiments/products in own field of research and in own settings.
Venue
The course will be convened on the campus of Korea University. No fee for registration.
Accommodation
Accommodation arrangements have been made in the Korea University Guest houses. Meals will be provided during course.
Arrival in Seoul
The city of Seoul is well connected with rail, road and air. Several international flights fly in Seoul directly.
AGENDA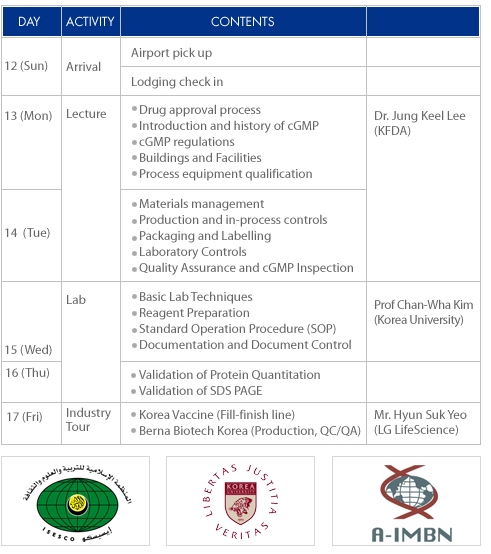
Chief Organizer
Prof. Can Wha Kim (cwkim@korea.ac.kr)
Key Instructor
Dr. Jung Keel Lee
Apply for click here (Application form Download) - * Attached file : Application form.doc
ISESCO and A-IMBN Training Course on Good Manufacturing Practices (GMP), Sept. 13 – 17, 2010, Seoul, Korea
2010/07/08
 back
back
Tel +82. 2. 872. 8016 Fax +82. 2. 882. 6702 Email kevin@a-imbn.org
Copyright(c) 2010 by A-IMBN. all rights reserved
